Could a tiny bacteria causing gum disease in the mouth lead to the development of Alzheimer’s pathology in the brain?
Scientists and researchers discovered a bacteria associated with chronic gum disease and linked it to the development of pathologic changes of Alzheimer’s disease. The study published in the journal Science Advances found byproducts of a chronic gum disease associated bacteria called Porphyromonas gingivalis in the brains of patients with Alzheimer’s (1). Shocking? Yes!
A mouse model confirmed that the bacteria could actually travel from the mouth to the brain. Once there, the bacteria secrete a toxin called gingipain which destroys neurons. Gingipain is a protease. The root ‘gingi’ comes from the association with gingivitis, inflammation of the gums largely responsible for tooth decay. The ‘pain’ part refers to the action of the protein just like papain, the active ingredient in meat tenderizer. Proteases cleave proteins just like a meat cleaver chops meat. The structure of the body, in large part, is scaffolded together with protein. The study investigators identified these toxic gingipain proteins
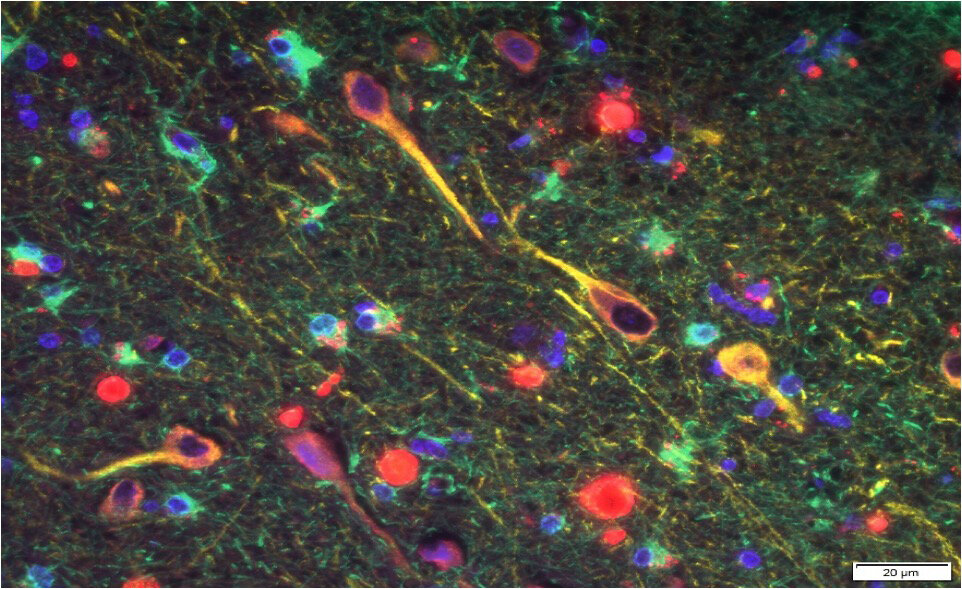
in the brains of humans with Alzheimer’s disease. The researchers also confirmed the gingipain proteins were directly toxic to neurons and displayed characteristic effects on tau (the other protein associated with AD). The production of amyloid beta (Aβ1-42) was increased in the regions of the mouse brains colonized with P. gingivalis.
Dr. Dale Bredesen’s ReCODE (Reversing COnitive DEcline) program taught us that amyloid beta, while undeniably harmful, is actually a protective response of the brain (2). Historical thinking was that since amyloid beta was associated with the development of Alzheimer’s, that it must be ‘the cause.’ It turns out nothing could be farther from the truth! Amyloid beta is the response, not the cause. We must find the reason why it’s being over-produced and eliminate that reason, not merely the response to it. Fortunately that’s how we’re accustomed to looking at things from the Functional Medicine perspective. Always look for the cause of the cause.
It turns out that multiple pharmaceutical corporations have spent billions and billions of dollars investigating new agents, many directed at amyloid beta, in hopes of curing Alzheimer’s. Almost a one, they have ended in failure. Worse yet, some actually caused the disease to progress more rapidly. Why? One reason is that amyloid beta is actually a protective response. Dr. Bredesen likens it to napalm (the jungle defoliant used in the Vietnam War). The amyloid destroys the invaders, albeit with a scorched Earth sort of tradeoff. The resulting wake of destroyed neurons is responsible for the cognitive decline (2). But the brain is limited in ways that it can respond to a threat which has breached the blood-brain barrier. Elsewhere in the body, infection would be dealt with directly by the white blood cells of the immune system. But the brain is inside a protected space and is forced to use other means. We now know one of the most important keys is to find the reasons why amyloid beta is being produced and reduce or eliminate those underlying causes. Again, the reason for our motto, Look Deeper™️.
The sad fact is that P. gingivalis is only one reason why the brain may be over-producing beta amyloid. And it turns out there are over 100 other reasons! Some of the reasons are from other infectious agents, be those bacterial as in the case of P. gingivalis, or viral in others. And the list goes on, and on! Fortunately, at Peak Health we have access to the specialized tools and testing to uncover these and other reasons. Those reasons will always be different from one individual to another. Our training has prepared us for dealing with such complexity. It truly is where the power of personalized medicine comes into play.
Just as there may be many causes for a single condition like Alzheimer’s disease, each of those causes may, in turn, result in other conditions. Understanding and dealing with complexity is why we find Functional Medicine such a fascinating paradigm for understanding an individual’s overall health. In the case of P. gingivalis we’ve explored its relationship with Alzheimer’s. However, it doesn’t end there. This bacterial pathogen, as well as others, are associated with the development of head and neck squamous cell carcinoma (3). P. gingivalis has also been associated with the creation of an inflammatory situation in the GI tract called dysbiosis (4) which creates repercussions throughout the body. The organism is also known to have strong ties to the development of rheumatoid arthritis (5). And most certainly, there are other associations yet to be discovered.
We will keeping a close watch for further developments with regards to P. gingivalis. There is already work underway to develop a small molecule inhibitor which could block the toxic effects of gingipain. In October 2018, a company named Cortexyme announced encouraging results from a Phase 1b clinical trial of COR388 which was shown to have positive trends on cognitive testing in patients with AD (6).
-
Porphyromonas gingivalis in Alzheimer’s disease brains: Evidence for disease causation and treatment with small-molecule inhibitors
- The End of Alzheimer’s: The First Program to Prevent and Reverse Cognitive Decline by Dr. Dale Bredesen.
- Gallimidi, Adi Binder, et al. “Periodontal Pathogens Porphyromonas Gingivalis and Fusobacterium Nucleatum Promote Tumor Progression in an Oral-Specific Chemical Carcinogenesis Model.” Oncotarget, vol. 6, no.
26, 2015, doi:10.18632/oncotarget.4209. - Olsen I, Lambris JD, Hajishengallis G. Porphyromonas gingivalis disturbs host–commensal homeostasis by changing complement function. Journal of Oral Microbiology. 2017;9(1):1340085. doi:10.1080/20002297.2017.1340085.
- Scher JU, Abramson SB. Periodontal disease, Porphyromonas gingivalis, and rheumatoid arthritis: what triggers autoimmunity and clinical disease?. Arthritis Res Ther. 2013;15(5):122.
- https://medicalxpress.com/news/2019-01-bacterial-pathogen-porphyromonas-gingivalis-contribute.html
Copyright 2019, Peak Health LLC